Advancing pharmaceutical processing, transforming the future, saving lives
15 Sep 2021

GEA and its partners are leading the way toward smaller, more flexible, continuous processing technologies that are transforming the future of pharmaceutical development and manufacturing.
The ability to deliver customized quantities of drugs to patients in need in a quick and efficient way — particularly with the increasing focus on precision medicines — is what the pharmaceutical industry has and always will strive for. Previously, however, the batch-based production of blockbuster oral solid dosage (OSD) forms dominated the industry so much that drug production companies were not incentivized to innovate or risk developing new manufacturing technologies. That was then, though, and this is now!
GEA has not only been helping pharmaceutical manufacturers to evaluate, develop and optimize continuous processing techniques to enable them to bring new products to market faster, the company has taken a proactive stance in the industry and spearheaded several collaborations and partnerships that have championed the implementation of continuous manufacturing (CM) to highlight its many advantages (see sidebar).
For example, back in 2015, the US FDA approved a pharmaceutical OSD form (ORKAMBI, Vertex Pharmaceuticals Incorporated) that had been developed and produced using GEA’s ConsiGma® CM platform for the first time. Further, approvals followed in 2016 (PREZISTA®, Janssen), 2017 (Verzenio, Eli Lilly) and 2018 (SYMDEKO, Vertex).
Furthermore, beyond its proprietary and multipurpose ConsiGma® 4.0 platform — designed to transfer powder into coated tablets in development, pilot, clinical and production volumes — GEA, GSK, Pfizer and G-Con Manufacturing formed a consortium to design and built a modular, autonomous manufacturing environment for continuous OSD form production. Presented with the award for Best Technologies Innovation during INTERPHEX 2015 and subsequently receiving an ISPE Facility of the Year Award (FOYA) in 2016 for Equipment Innovation, this first-of-a-kind portable, continuous, miniature and modular (PCMM) system shortens the timelines involved in tablet production.

Transforming the way the Pharmaceutical Industry makes Tablets (courtesy of Pfizer)
By miniaturizing the equipment, the whole continuous process can be positioned within in a small-footprint facility that can be shipped to any location in the world and quickly assembled. Once up and running, the system has the capability to transform powders into uncoated tablets in minutes, which can take weeks or months with current technology. With a 60–70 percent smaller space requirement than a traditional set-up and lower energy requirements, CM also offers environmental benefits and a lower carbon footprint.

Portable pharmaceutical production facilities delivers medicines to patients faster (courtsey of Pfizer)
More recently, while evaluating ways to further increase the value of their products and processes, Janssen Belgium realized that they were reaching the operational limits of their batch manufacturing technologies. As a result, the company embarked on a strategically ambitious project to implement an end-to-end CM line. “The unique advantages of introducing an end-to-end platform will help us to develop novel products that would not otherwise be possible,” commented a Janssen spokesperson. What’s more, for their aspirational approach to advancing CM, Janssen received the 2020 FOYA in the Process Innovation category.
Now, increasingly gaining momentum in the drug manufacturing sector as the new normal approach to OSD form production, CM can help to transform today’s pharmaceutical business processes into a future market environment driven by global megatrends, such as ageing populations, growing middle classes in emerging markets, increased patient safety, cost reductions in public and private healthcare and increased regulations.
Breakthrough work was done by MSD (a tradename of Merck & Co., Inc.), for example, who, having subscribed to a comprehensive virtual reality training program, undertook an extended trial run to assess the robustness of GEA ConsiGma® CDC 50 Continuous Direct Compression system. By the end of the 120-hour test, more than 15 million tablets had been made using approximately 6200 kg of raw material in a single production run.

GEA’s ConsiGma CDC at MSD, Cramlington, UK (courtesy of MSD)
Benchmarking against a typical batch process, producing the same quantity of tablets would have required 10 separate campaigns and taken a team of operators working in parallel for 5–7 days. During the overall run, the yield loss was minimal, measuring just 0.05 percent, and less than 0.5 percent of the tablets were out-of-specification.
Importantly, final analysis indicated that the campaign length could be increased even further and run for longer. As such, it was shown that CM is conclusively able to reliably, flexibly and compliantly produce high-quality drug products in response to any production demand.
Not that batch systems are going away anytime soon, but the use of small-scale CM systems has already prompted one of the most noteworthy changes in the pharmaceutical industry. Flexible development options now facilitate the commercial manufacturing process and enable greater process understanding to be achieved with smaller quantities of material.
The ability to obtain more data from less product during development and eliminate the cost and risk of batch-based “scale-up” has inspired the introduction of small-scale, continuous equipment that can process small quantities of material during R&D while also being able to operate for variable lengths of time to match market demand during commercial production.
In addition, regulators are increasingly supportive of CM and manufacturers are recognizing that current quality costs are disproportionately large compared with other industries, wherein the production, detection and removal of out-of-specification product is vanishingly small.
Recently, for instance, Pfizer has announced that it’s investing £10 million (€11.7 million) to speed up medication production in the light of the COVID-19 pandemic. The company says that the coronavirus has highlighted the need to create new vaccines and medicines at pace and scale. Such pressures have put the costs, risks and timelines associated with traditional batch-based development and manufacturing under scrutiny.
Shortening manufacturing processes and reducing variability are key factors in enabling clinical trials to progress and could drastically speed up the delivery of life-changing treatments to patients around the world.”– Pfizer spokesperson
– Pfizer spokesperson
The investment includes a PCMM to rapidly develop tablets for clinical trials around the world. “The technology means you increase the module’s hours of use to scale-up manufacturing from thousands of tablets to hundreds of thousands or even millions when needed,” said the company representative, adding: “Working on both physical and virtual production side by side improves efficiency by cutting down the amount of materials needed and the time it takes to develop each formula.”
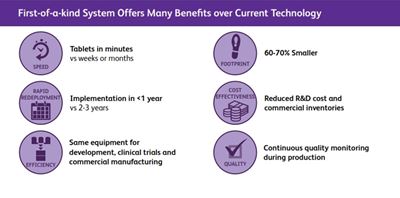
First-of-a-kind system offers many benefits (courtesy of Pfizer)
Here again, GEA is at the forefront of innovation: to enhance the customer experience, the company now offers an online platform of process simulations, digital twins, augmented/virtual reality systems and remote support solutions that provide the perfect environment for digitally enabled project execution.
Specific to the pharmaceutical industry’s needs, GEA is also addressing the drug development phase with probes and PAT instruments to better design processes at an early stage. With advanced analytical tools and the ConsiGma® 4.0 R&D product line, Quality by Design (QbD) principles can be applied all the way from early stage product development through to commercial-scale manufacturing, enabling significant time savings and faster process transfers.
Pfizer estimates that 70 percent of its small molecule, OSD medicines will be manufactured on PCMM by 2029. GEA and forward-thinking companies such as Vertex, Pfizer and Janssen are keen supporters and drivers of this innovative manufacturing technology. And whereas many companies talk about CM, these GEA customers have actually implemented it and are benefiting from the advantages it offers. Supported by the US FDA and approved for the development and production of novel oral solid dosage forms — many of which were not possible using traditional approaches and equipment — CM is not just a concept; it’s real and already delivering on its promises.
Pfizer, GSK, GEA and G-CON Manufacturing - Getting Medicines to Patients Faster
Courtesy of Pfizer

